
DO YOU HAVE CANDIDA OVERGROWTH?
If you are experiencing chronic symptoms ranging from Brain Fog, Intestinal Discomfort, Joint Pain to Fatigue, but your doctor is at a loss to explain the reason, Candida Overgrowth could be to Blame!
What is Candida?
Humans host a complex microbial ecosystem composed of bacteria, fungi, viruses, protists and archaea – each playing key and interconnected roles in maintaining health and supporting biological functions. The gut microbiome varies greatly between individuals, both in size (total microbial load) and composition (species diversity), due to differences in physiology, diet, lifestyle, and environment.
Candida is a yeast-like fungus that naturally lives on and inside the human body – primarily in the mouth, gut, skin and vaginal tract – without causing harm under normal circumstances (1). However, Candida can become opportunistic under certain conditions and cause infection – when the normal balance within the microbiome is disrupted or the host’s immune system is weakened (1). The main drivers of candida overgrowth include poor diet, antibiotics, stressful lifestyle, alcohol, high blood sugar, contraceptive hormones, immune suppression and gut disrupting drugs.
Over 150 Candida species have been recognized to date. But Candida Albicans is the most frequently associated with HUMAN INFECTIONS.
It is a dimorphic Gram-positive yeast, not acid-resistant, saprophyte (fungus that lives on dead or decaying matter) in 40–75% of healthy humans (28, 29). And is responsible for over 80% of human yeast infections (28,30).
Among humans – Candida Albicans is the most studied and clinically relevant – an overgrowth of which affects millions of people; many of whom are unaware of the cause of their symptoms. In fact, it is estimated that one in three people suffer from yeast overgrowth due to Candida Albicans! Candida albicans is a common opportunistic yeast that causes oral thrush, vaginal infections, and gut issues!
The genus name Candida is attributed to the traditional “white robes” worn by Roman candidates (candidatus) running for public office. The term albicans is derived from another Latin word albico/albicatus, which means “to be white” or “verge on white.” In essence, the term Candida albicans is redundant meaning “white to be white” and is associated with white color [2].
What is Candida Overgrowth or Pathogenicity?
Candida overgrowth happens when the natural balance between Candida yeast and beneficial bacteria in the body is disturbed, allowing candida to multiply excessively and cause infection. This can occur in various parts of the body such as the gut, mouth, throat, or genital area.
When Candida overgrows, it disrupts the normal microbial balance and can lead to symptoms and infections collectively known as Candidiasis.
In most individuals with a healthy immune system, C. albicans is a harmless microbe that exists in harmony with other members of the gut flora that keep candida under control.
Candida begins to overgrow when the environment in the body changes due to the following factors:
- Repeated use of broad-spectrum antibiotics (kills both good and bad bacteria creating room for fungus to grow)
- Changes in the host immune system (e.g., during stress, poor diet and lifestyle, infection by another microbe, or immunosuppressant therapy)
- Variations in the gut environment (e.g., shifts in gut pH, gastritis, or nutritional deficiencies) and,
- Conditions such as diabetes, HIV/AIDS, chemotherapy, organ transplantation
- Damaged physical barriers (damaged mucosal barriers such as mouth, gut, genital surfaces due to trauma, chemotherapy, or medical devices for example catheters)
These conditions favor the trigger of fungal proliferation causing Candida to grow out of control, invade deeper tissues and turn into an aggressive pathogen causing wide range of infections (1).
These infections range from:
- superficial mucosal and dermal infections, such as oral thrush, diaper rash or skin rash, and vaginal yeast infections (75% of women will have yeast infection or UTIs at least once in their lifetime),
- to more serious blood/organs related and deep tissue infections with sizable mortality rates (approaching 47% in some cases) (1).
While women are more susceptible to vaginal or genital yeast infections (also known as UTIs in lay term), men and kids can also be infected.
Certain factors, such as prolonged antibiotic use, increase the risk of candida overgrowth for both men and women. Prolonged Antibiotics use promotes yeast (fungal) infections, including gastrointestinal (GI) Candida overgrowth and penetration of the GI mucosa making the host more susceptible to yeast infections. Candida albicans may also play a role in the persistence or worsening of some chronic inflammatory bowel diseases (IBD) (5,6).
Many of the illnesses and symptoms that plague men and women and children today – from fatigue, bloating, and weight gain to prostates, brain fog, arthritis, allergies, ear infections and depression can be traced back to surprising factor – an overgrowth of yeast called Candida Albicans (5,6).
In addition to these concerns, candida has also been linked to arguably more serious and debilitating illnesses such as autoimmune diseases like multiple sclerosis, fibromyalgia, autism, rheumatoid arthritis, lupus and mental illness (5).
Infection caused by Candida are especially serious in immunocompromised individuals (such as those with diabetes, nutrient deficiencies, AIDS/HIV or those undergoing anticancer chemotherapy/radiation immunosuppression therapies) and healthy people with implanted medical devices such as catheters, pacemakers, prosthetics, heart valves, dentures etc. (1,6).
Candida’s ability to shift from a harmless commensal to an aggressive pathogen exemplifies its opportunistic nature – it takes advantage of weak immune system or disrupted microbial imbalance to cause a disease.
Factors that lead to Candida Overgrowth
- Diet high in sugar and refined carbs – Candida thrives on sugar.
- Repeated Antibiotic use – Antibiotics kill beneficial and non-beneficial bacteria (not fungi), that normally keep candida in check. This disrupts the microbial balance allowing the yeast to overgrow.
- Low stomach acid (Impaired Upper Digestion) – impacts gut microbiome and again makes the environment favorable for candida overgrowth
- Weakened Immune System and Health conditions – Conditions such as HIV/AIDs, diabetes, cancer, chemotherapy, and use of immunosuppressive drugs increase susceptibility to candida
- Excess alcohol consumption – Excess alcohol can lead to leaky gut, change gut flora and thus act as a fuel source for candida overgrowth.
- Estrogen Dominance – Changes in hormone levels particularly estrogen, can contribute to yeast overgrowth.
- Chronic Stress – Chronic physical or psychological stress raises cortisol levels, which suppresses the immune system and raises blood sugar – both factors that fuel Candida overgrowth
- Oral Contraceptives and Hormone Therapy – Birth control pills and estrogen replacement therapy can promote Candida growth by disrupting hormonal and microbial balance.
- Other factors – Smoking, poorly maintained dentures, pregnancy, nutritional deficiencies, and chemical exposures can also predispose to candida overgrowth.
How does it enter your body?
Fungi or candida infect the body through several portals of entry. The first exposure to fungi that most humans experience occurs during birth, when they encounter the yeast while passing through the vaginal canal of their mother. During this process the fungus colonizes the buccal cavity and portions of the upper and lower gastrointestinal tract of the newborn, where it maintains a life-long residence as a commensal or friendly yeast) (3).
How Candida destroys your gut?
Candida destroys your gut by forming a biofilm and inducing morphogenesis. A major virulence attribute of Candida albicans is its ability to form Biofilms. Biofilms are densely packed communities of cells adhered to a surface. Fungi make biofilms to protect themselves from host’s immune system and therapeutic antifungals (6).
Recent estimates by the National Institutes of Health (NIH) indicate that pathogenic biofilms are responsible, directly or indirectly, for over 80% of all microbial infections (6).
When candida multiplies excessively, it can shift from a harmless yeast form into a filamentous (root like hyphal) form (1,6). These hyphae/Candida roots can penetrate the gut and firmly anchor themselves into the intestinal lining of your gastrointestinal tract or gut, causing the intestinal barrier to become more permeable, thus widening the gaps between them. This physical damage is known as “leaky gut” or “leaky gut syndrome.”

When the wall that separates your gastrointestinal tract from your bloodstream becomes permeable, the fungus and its toxic by-products can seep through the intestinal lining into your bloodstream causing inflammation. This inflammation further weakens the gut barrier and perpetuates increased permeability, leading to more substances escaping into the blood, which can cause systemic inflammation and even food sensitivities or allergies.
The inflammation can show up on your skin in the form of acne, dandruff, eczema, or dry and itchy skin on an acute level. Once the invasion takes place, candida and its mycotoxins (toxins produced by candida are known as mycotoxins) can accumulate in the body, causing cellular disruption within the immune system and your organs and tissues. Mycotoxins so severely debilitate the body that the individuals with weak immune system easily fall prey to far more serious diseases like multiple sclerosis, rheumatoid arthritis, ulcerative colitis, and even schizophrenia (5).
Candida overgrowth often develops alongside microbiome imbalance. As gut permeability increases, gut immunity worsens, and beneficial bacteria decline. Candida flourishes under such opportunistic conditions creating a vicious cycle of gut dysbiosis and Leaky gut (1,6).
Candida albicans can form biofilm on mucosal surfaces, such as those coating the oral (dentures) and vaginal epithelia, and implanted medical devices, such as prosthetics, heart valves, pacemakers, and catheters, which can seed systemic infections in humans (1,6).
Signs and Symptoms of Candida Overgrowth
- Digestive issues – Gas, Bloating, Constipation or Diarrhea
- Recurring Bladder infections – Urinary Tract Infections (UTIs), Vaginal infections, Rectal itching or Vaginal itching
- Intensified sugar and refined carbohydrate cravings
- Difficulty concentrating, poor memory, lack of focus, ADD, ADHD, or brain fog
- Feeling tired and worn down or suffering from fatigue or fibromyalgia
- Irritability, mood swings, anxiety, or depression
- Skin issues including dandruff, dry and itchy skin, eczema, psoriasis, hives and rashes, rosacea and acne
- Skin and nail fungal infections – athlete’s foot, ringworm, and toenail fungus
- Autoimmune diseases such as Hashimoto’s thyroiditis, rheumatoid arthritis, lupus, psoriasis, or multiple sclerosis
What is Candidiasis?
Candidiasis refers to Candida Overgrowth! It is caused due to an excessive proliferation of C albicans and other Candida species under certain conditions; and is the most common fungal/yeast infection in humans.
Candida Albicans is the most common cause of Candidiasis!
Candidiasis may be classified as:
- Superficial and
- Systemic (Deep)
Superficial candidiasis may involve the infection at – epidermal and mucosal surfaces, including those of the oral cavity, pharynx, esophagus, intestines, urinary bladder, and vagina (4).
The major portals of entry for deep (or visceral) candidiasis are – the alimentary tract and intravascular catheters. The major organ sites affected or involved in deep or visceral candidiasis are kidneys, liver, spleen, brain, eyes, heart, and other tissues (4).
Types of diseases and conditions associated with Candida Overgrowth:
1.Oral Candidiasis – (oral thrush) – This is a fungal infection in the mouth, commonly known as oral thrush. It is characterized as acute pseudomembranous candidiasis, but erythematous (a redness of the mucous membrane, often indicating inflammation or irritation) forms also exist (21). Oral Candidiasis is most common in people who wear dentures or have nutrient deficiencies.
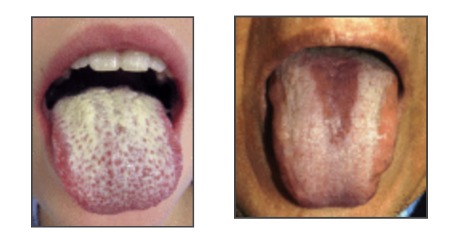

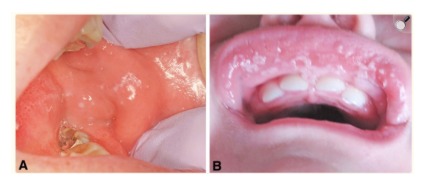

While Candida albicans is by far the most common species associated with oral thrush, at least seven other species within the Candida genus have been attributed to the disease in the oral cavity: C. glabrata, C. guillermondii, C. kruesi, C. lusitaniae, C. parapsilosis, C. pseudotropicalis, C. stellatoidea, and C. tropicalis (24).
As estimated 30–60% of healthy adults carry Candida species within the oral cavity. In healthy patients, the patient’s immune system and normal bacteria flora inhibit candida growth. Candidal species cause oral candidiasis when a patient’s host immunity becomes compromised. Overgrowth of the fungus then leads to the formation of a pseudo membrane.
Risk factors include – immunosuppression due to diabetes, use of dentures, topical steroid use (in the form of inhalers, gels, or rinses) and decreased salivary flow, malnutrition, high intake of sugar, vitamin deficiencies (iron deficiency anemia, folic acid, thiamine, riboflavin, vitamin B12 deficiency), and recent antibiotic use (23, 25).
A patient’s oral Candida infection can often lead to GI involvement and subsequent candidal diaper dermatitis (skin rashes) for Candidal species thrive in moist environments. Alternatively, neonates (newborns) and infants may contract the disease through mother’s breasts that are colonized with candida – when breastfeeding.
- Treatment focuses on the type of Candida species present and is targeted to the extent of the patient’s involvement and degree of immunosuppression.
- Topical antifungal therapy and oral hygiene measures are usually sufficient to resolve mild oral candidiasis.
- Systemic antifungal therapy is usually reserved for patients who are intolerant to topical therapy, and those at increased risk of developing systemic infections (21, 24).
2. Vulvo Vaginal Candidiasis (VVC) – This is a common yeast infection among women, caused in the Vagina.
Approximately 70% of women report having had candidal vulvovaginitis (VVC) in their lifetime, and an estimated 8% of women suffer recurrent candidal vulvovaginitis (RVVC) (8,9). The most common responsible pathogen is C. albicans, accounting for 90% of cases, with most of the remaining cases caused by Candida glabrata.
Symptoms include – itching and redness in private parts, thick, white and clumpy vaginal discharge that may resemble cottage cheese, pain during urination (dysuria) or intercourse, swelling and irritation in vagina, soreness or burning sensation in the vulva and vaginal areas (8). It is often triggered by excessive use of antibiotics, increased estrogen levels (e.g. high estrogen oral contraceptives, hormone replacement therapies, pregnancy), uncontrolled diabetes mellitus, sexual activities and tight-fit clothing, hormonal changes, or immune suppression (8,10,11). Vaginal infections can colonize neonates (new borns) as they pass through the birth canal.
Although not associated with any mortality, VVC and RVVC are associated with considerable morbidity. Symptoms of vaginitis can cause substantial distress, resulting in time lost from work and altered self-esteem (7,12). Thus, it is not surprising that vaginal complaints are the most common reason for gynecological consultation. Among the many causes of vaginitis, VVC is the second most common after bacterial vaginosis, and it is diagnosed in up to 40% of women with vaginal complaints in the primary care setting (7).
Note – VVC is different than UTI. VVC is a fungal infection whereas UTI is a bacterial infection both occurring in women. However, VVC is sometimes referred as UTIs (urinary tract infections) in lay terms because VVC and UTI can have overlapping symptoms.
However, Men too can get a Urinary Tract Infection (UTI) from Candida, although it is much less common than bacterial UTIs. Candida yeasts can cause UTIs by infecting the urinary tract through antegrade (from the bloodstream) or retrograde (via the urethra) routes. These fungal UTIs are also known as candiduria and are most often seen in men with risk factors like antibiotic use, diabetes, catheter use, or immunosuppression (7).
Men are also susceptible to another type of candida infection – called Balanitis Candidiasis. Balanitis is an inflammation of the “head of the penis” (glans). This can be painful and make urinating more difficult. If left untreated, it can cause swelling and pain in the glands, as well as weakness and fatigue (7). Diabetes can increase your risk for Balanitis!
Symptoms of Balanitis are:
- Local burning, and itching accompanied by pain
- Thick, Foul-smelling Discharge
- Inflammation and redness of the head of the penis
- The clinical features include – redness and solid bumps with or without small pus-filled bumps of the area – with surrounding redness and erosion (15).
Some people experience inflammation of both the head and the foreskin of the penis, known as balanoposthitis (7). Without effective treatment, balanitis can lead to scarring of the foreskin and can cause adhesions on the penis. It may prevent you from being able to pull your foreskin back, making proper hygiene impossible. Candida species. are responsible for 30 to 35% of all cases of infectious balanitis (13,14,15) and for up to 54% for diagnoses based solely on clinical examination (15).
3. Esophageal candidiasis – also known as esophageal thrush, is a serious yeast infection affecting the esophagus caused by Candida. It is common in immunocompromised individuals and can lead to pain when swallowing and lesions seen on endoscopy. Other symptoms include abdominal pain, heartburn, weight loss, malnutrition, diarrhea, nausea, vomiting, ulceration and hemorrhage (16,17,18).
Immunosuppressed patients at risk for esophageal candidiasis include GERD (Gastro Esophageal Reflux Disease), HIV positive and AIDS patients, chemotherapy patients, patients with radiation to the neck region, antibiotic therapy, patients on chronic systemic or topical inhaled corticosteroids, diabetes mellitus, adrenal insufficiency, and advanced age (16,17,18). Studies have shown that the use of proton-pump inhibitors is also a strong risk factor for esophageal candidiasis in immunocompetent patients. Some studies show that smoking tobacco also correlates with developing esophageal candidiasis (16,18,19,20).
By far the highest risk factor for developing esophageal candidiasis is impaired cell-mediated immunity. Due to impaired cell-mediated immunity, the esophageal epithelial layer is susceptible to infection and colonization by candida. The candida proliferates and adheres to the esophageal mucosa forming white-yellow plaques. The plaques can be seen on upper endoscopy and do not wash from the mucosa with water irrigation. These plaques can be found diffusely throughout the entire esophagus or localized in the upper, mid, or distal esophagus (16,18).
Since candida is a normal oral flora that proliferates in immunocompromised states of health, one way of decreasing the risk of esophageal candidiasis is to improve health conditions that can cause immunosuppression. Decreasing the use of antibiotics, systemic steroids, and the proper use of inhaled steroids can also be used to limit the risk of esophageal candidiasis. Prophylactic fluconazole may be necessary for patients that have recurrent infections (16,18).
4. Skin and Nail Infections – Overgrowth of candida on the skin can cause rashes, itching, and discomfort, particularly in warm, moist areas such as armpits and groin. It can also affect the nails, leading to fungal nail infections such as toenail fungus or athletes’ foot.
Skin is a barrier structure exposed to both commensal (friendly and harmless) microbes and pathogens (harmful). C. albicans is a dimorphic fungus that grows as a commensal on barrier surfaces like skin but can also become pathogenic [19]. The symptoms of skin and mucosal membrane infection are generally mild. C. albicans tends to occur in skin folds, such as the armpit, groin, under the breast, around the anus and in nail folds, causing clinical skin diseases in the form of onychomycosis (nail infection), paronychia (skin infection around the nail), and diaper rash (27).
5. Invasive Candidiasis – This is a serious infection that occurs when candida enters the blood stream and spreads to internal organs, such as the heart, brain, eyes, bones, and joints. This condition most often affects people who are hospitalized or have weak immune system and can be life threatening. C. albicans is a leading cause of hospital-acquired infections, it accounts for 15% of all sepsis cases and is the cause of 40% of bloodstream infections in clinical settings (1,42).
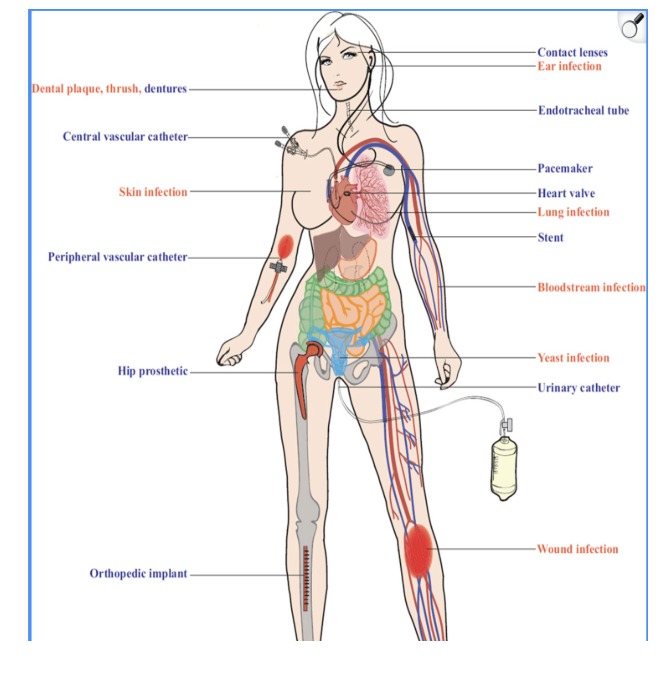
C. albicans is the predominant fungal species isolated from medical device infections, including urinary and central venous catheters, pacemakers, mechanical heart valves, joint prostheses, contact lenses, and dentures (43). See Figure 2 below.
C. albicans forms highly structured biofilms composed of multiple cell types. Once a biofilm is formed on an implanted medical device, it acts as a reservoir for pathogenic cells, is highly resistant to antifungal drugs and the host immune system and has the potential to seed disseminated bloodstream infections (known as candidemia) that can lead to invasive systemic infections of tissues and organs.
Each year in the United States, over 5 million central venous catheters are placed and currently – even with recent improved clinical approaches – biofilm infection occurs in over 50% of these catheters (1,42). With an estimated 100,000 deaths and $6.5 billion in excess expenditure annually in the United States alone, these infections have serious health and economic consequences (1).
Additionally, as these fungal biofilms are largely resistant to known antifungal drugs, the current standard of care to treat these infections involves the removal of the colonized medical device, oftentimes through surgery, combined with administration of high doses of antifungal agents (44,45). Removal of some of these devices (e.g. artificial heart valves and joints) can be costly and, in some cases,dangerous to the patient, and the administration of high doses of antifungal agents (typically given intravascularly), can result in further complications, including kidney and liver damage (42,44). Oftentimes, these treatments are not even possible, as many critically ill patients are unable to tolerate them, leaving these patients with few available options and underscoring the need to find better therapeutic and diagnostic therapies to combat these biofilms.
Saccharomyces cerevisiae, or baker’s yeast, rarely causes infections in humans and is generally not considered a pathogen. In a few case studies, however, S. cerevisiae has been implicated in catheter-associated infections with mixed-species biofilms in patients in intensive care units (ICUs), and it is able to form a thin biofilm consisting of round, budding yeast-form cells and pseudo hyphal cells in vitro (28).
Candida Albicans is the predominant fungal species isolated from the following medical devices as shown in the picture below (Fig 2).
- Dentures
- Central vascular catheter
- Peripheral vascular catheter
- Hip prosthetic
- Orthopedic implant
- Pacemaker
- Heart valve
- Stent
- Urinary catheter
- Endotracheal tube
- Other infections include – ear infection, lung infection, bloodstream infection, yeast infection or UTIs, skin infection, dental plaque, oral thrush, and wound infection.
Fig 2 - This picture highlights the areas of female body directly colonized by a biofilm through the use medical devices – associated with Candida albicans biofilm infections (Ref – 1).
6. Gastrointestinal disorders – Overgrowth of Candida in the gut has been linked with gastrointestinal diseases including Irritable bowel disease (IBD), Crohn’s disease, ulcerative colitis, gastric ulcers and duodenal ulcers. It can also cause symptoms like bloating, diarrhea, abdominal pain, cramps, and nausea (26).
Further, results from animal models argue that Candida colonization delays healing of inflammatory lesions and that inflammation promotes colonization (26). These effects may create a vicious cycle in which low-level inflammation promotes fungal colonization and fungal colonization promotes further inflammation. HOW?
Both inflammatory bowel disease (IBD) and gastrointestinal Candida colonization is associated with elevated levels of the pro-inflammatory cytokine IL-17. Therefore, effects on IL-17 levels may underlie the ability of Candida colonization to enhance inflammation (26). Also, an antibiotic treatment often leads to GI tract inflammation, which may perturb the resident bacterial community, allowing C. albicans to colonize. Furthermore, in the GI tract, C. albicans encounters and responds to varying features of the physical environment such as pH, oxygen levels and nutrient levels (47). C. albicans also responds to secretions produced in the GI tract such as bile (48). These findings argue that C. albicans is well adapted for growth in the GI tract.
Because Candida is a frequent colonizer, these effects have the potential to impact many people by exacerbating inflammation and creating a vicious cycle of colonization and inflammation – ranging from gut issues to whole body issues.
In simple words – The presence of inflammation alters bacterial colonization and the activities of the host, creating conditions that favor both high level Candida colonization and exacerbation of disease like IBD.
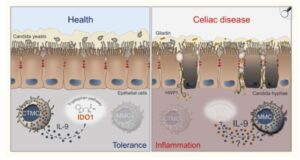
7. Candida and Gluten Intolerance –
The protein HWP1 (Hyphal wall protein 1) found on the surface of Candida Albicans closely resembles gluten proteins (alpha-gliadin and gamma-gliadin). When Candida overgrows and attaches to the gut-wall using HWP1 protein, the immune system attacks this protein, but because of its similarity to gliadin (gluten protein), may also develop reactivity to gluten, leading to gluten intolerance or even autoimmune reactions such as celiac-like conditions. This phenomenon is known as molecular mimicry (49).
Furthermore, Candida overgrowth has been proposed as a cause for leaky gut, where toxins and undigested foods leak into the blood stream, leading to allergic reactions and inflammation. This makes the digestive tract more reactive to gluten and other proteins. As a result of this cross reactivity, candida may trigger gluten sensitivities by confusing the immune system, especially when gut health is compromised by candida overgrowth (50). Conversely, those with celiac disease or gluten sensitivity may be more prone to candida overgrowth because compromised gut health (due to antibiotics overuse, diabetes, pH shift, nutrient deficiencies, stress, etc) increases susceptibility to fungal imbalance as shown in the picture below (49,50).
In such cases, addressing candida overgrowth can sometimes alleviate gluten sensitivities, especially in non-celiac cases, by reducing gut inflammation and improving immune regulation. Also, for those experiencing persistent digestive symptoms, fatigue, or skin problems, considering both candida and gluten intolerance is important for accurate diagnosis and effective treatment (50).
Schematic depiction of the interactions between Candida, MCs and IL-9 in celiac disease (Ref 50).
8.Candida and Iron Deficiency – candida may make someone iron deficient by competing for iron, impairing iron absorption, and triggering host responses that further lower available iron stores.
Candida overgrowth related iron loss is particularly important to consider in individuals with chronic gut symptoms, unexplained anemia, or recurrent candida infections. Iron deficiency may manifest as fatigue, weakness, pale skin, and other symptoms.
Yes, candida overgrowth can contribute to iron deficiency through multiple mechanisms:
- Hepcidin release – The presence of candida stimulates the liver to produce antimicrobial peptide hormone called – Hepcidin – a hormone that blocks the release of iron from intestinal cells and storage tissues. Hepcidin reduces the release of iron from liver stores and decreases iron absorption from the gut to limit iron availability to pathogens. This action decreases circulating and stored iron, lowering ferritin levels (52).
- Intestinal disruption – Overgrowth of Candida can damage the gut lining, impairing iron absorption from digested food which is essential for maintaining ferritin stores. This damage decreases how much iron enters the bloodstream and can lower iron storage over time; especially if the gut microbiota is also imbalanced (51,52).
- Alteration of Microbiome – Antibiotics or other disturbances enable candida overgrowth, further disturbing iron homeostasis by impacting microbiota that might otherwise help in processing and absorption of iron (51,52).
- Nutrient Immunity – Iron homeostasis and bioavailability to either the host or invading pathogens is tightly regulated through coordination of hepcidin, iron binding proteins and several enzymes involved in iron metabolism (51,53,54). The process of sequestering iron by the host and restricting its bioavailability to invading pathogens is called nutritional immunity. During infection, immune cells surround candida as in systemic infections, sequestering iron to make it less accessible to the pathogen, which also reduces its availability for human physiological needs (51).
- Siderophore production – Candida has strong affinity for iron and produces specialized iron-binding proteins called siderophores, which scavenge iron from the host, making less iron available for absorption in the gastrointestinal tract (52).
- Multisource Uptake – It possesses multiple high-affinity iron uptake systems that allow extraction of iron from transferrin, hemoglobin, ferritin, and siderophores produced by other microbes within the host (51,52).
- Cycle of iron deficiency and candida – Low ferritin and iron deficiency can impair immune function, making it easier for candida to overgrow. Meanwhile, the overgrowth perpetuates ferritin depletion by consuming iron and triggering hepcidin production, creating a self-reinforcing cycle. Managing ferritin deficiency in candida overgrowth requires not only iron supplementation when needed but also addressing the underlying fungal imbalance to break this cycle (51).
9.Candida and Gut pH levels – Candida albicans can grow in acidic or alkaline environments, but its biology and pathogenicity shift depending on the gut’s pH. In an acidic environment, candida mainly exists as a relatively harmless yeast form. However, when the environment becomes more alkaline or neutral, candida can switch to its pathogenic fungal (hyphal) form, which is associated with overgrowth and disease symptoms in the host.
Alkaline or neutral environments – A study
(55) reports that C. albicans can actively alter the pH of its environment and induce it’s switch to the hyphal form, which is more aggressive and can invade tissue. The change in pH is caused by the release of ammonia from the cells produced during the breakdown of amino acids. This phenomenon is unprecedented in a human pathogen and may substantially impact host physiology by linking morphogenesis (candida’s switch from harmless to pathogenic form), pH adaptation, carbon metabolism, and interactions with host cells, all of which are critical for the ability of C. albicans to cause disease (55).
One environmental factor to which microorganisms must respond is extracellular pH. In the human body, pH can vary widely, from highly acidic (pH ~2) in the stomach to mildly acidic (skin and vagina), to neutral (bloodstream and parts of the gut), and even alkaline (some parts of the gut). Candida albicans, thrives in most of these sites and is highly tolerant to a wide range of environmental pH conditions, from pHs of <2 to pHs of >10 (55). albicanshas a remarkable ability to alter extracellular pH, creating a neutral environment from either acidic or alkaline starting conditions, with changes in pH from 4 to >7 in less than 12 h (55). The rise in pH induces hyphal morphogenesis, a key virulence trait of candida species, and is correlated with release of volatile ammonia from the cells.
Thus, C. albicans effectively auto induces morphogenesis under these conditions. The rise in pH is associated with the release of ammonia, a highly basic compound, as has been observed in other fungi (55).
Regarding the oral cavity, the constant mechanical action of saliva creates a true challenge for oral pathogens as it hinders their adhesion to oral tissues (56). It seems likely that, similar to other pathogenic fungi (Göttlich et al., 1995), C. albicans lipases can increase cell adhesion capacity, probably by enhancing the hydrophobicity of Candida cells following the lipase‐dependent release of fatty acids. This scenario appears to take place in patients with diabetes mellitus, whose reduced salivary flow, lower pH, and elevated levels of glucose make the oral cavity the perfect microenvironment to allow the transition from commensal to pathogenic yeast (56).
In summary, Candida’s growth, form, and immune interactions are tightly linked to the pH of its environment (57). Acidic pH tends to keep candida less virulent and more exposed to immune clearance, while alkaline pH encourages its more harmful fungal form and overgrowth.
It is important to note that stomach should be highly acidic with pH between 1 to 3 and intestinal tract should be slightly acidic (with a pH of 4 to 6.5) to avoid alkaline environment that allows candida to flourish. Additionally, a healthy vagina is naturally acidic, with a pH of 4.5 or lower, which is hostile to candida growth.
Restorative strategies for increasing the acidity of stomach:
- Consume Apple Cider Vinegar (ACV): The acid and enzymes in ACV can help kill excess yeast and support a healthy intestinal pH.
- Eat Probiotic Foods: Fermented foods like plain yogurt, kimchi, sauerkraut, and Kefir contain beneficial bacteria like Lactobacillus acidophilus that produce lactic acid and acetic acid, which help restore the correct level of acidity in the gut. Probiotic supplements can also be used.
- Cut out sugar: Candida thrives on sugar, so reducing your intake of refined carbohydrates, sugary fruits, and alcohol is critical to starving the yeast.
- Incorporate gut-friendly foods: A healthy diet that includes non-starchy vegetables, lean proteins, and healthy fats is essential for a balanced gut microbiome.
- Garlic supplements: Garlic has antimicrobial properties and may help reduce harmful bacteria that can disrupt vaginal pH balance.
- Maintain proper oral and vaginal hygiene: Avoid mouthwash, if you wear dentures clean them properly on daily basis. To maintain vaginal hygiene, avoid douching, avoid scented feminine products, and harsh soaps, as these can disrupt the vaginal microbiome and pH balance. Wearing breathable cotton underwear is also recommended.
What Functional Tests help in diagnosing Candida?
Testing is essential to differentiate the root cause of symptoms and ensure appropriate effective treatment. Following are the primary functional tests helpful in identifying candida overgrowth:
- Culture and Microscopy – Superficial candidiasis (mouth, vaginal, esophageal) is diagnosed by microscopic examination and culture of a sample from the affected area.
- Gut Microbiome test – also known as GI MAP test – involves stool analysis that helps in identifying the levels of candida present and the type of candida species present in the GI tract.
- Antigen or antibody test – This is a Blood test that measures the levels of antibodies (IgG, IgA, IgM) against Candida Albicans in the blood. Elevated levels may indicate a current or past infection.
- Urine Organic Acid test (OATS) – analyzes a urine sample to check for the presence of fungal metabolites
- Blood tests and PCR – Invasive candidiasis is diagnosed with blood cultures or PCR-based methods (e.g., T2Candida panel, which can identify candida rapidly and with high sensitivity in blood). This is the most definitive test for diagnosing a systemic (bloodstream) infection.
Treatment and Management
Candida treatment and management involve a combination of lifestyle and dietary adjustments along with some natural remedies. The specific approach depends on the type, severity, and location of the Candida infection.
Here are few steps to follow:
- Candida detox through anti-candida diet and adjusting lifestyle measures
- Herbal Antifungals
- Probiotics push back
- Pharmaceutical antifungal drugs
How to do Candida Detox?
- Avoid foods that feed unhealthy candida levels in your body – Refined sugar, refined carbohydrates, Trans fat or seed oils, dairy products, excessive amounts of animal protein, excess caffeine and alcohol, gluten (wheat, barley, rye), GMO Corn and Soy, and artificial sweeteners, sugary beverages and condiments that contain hidden sugar.
- Incorporate these foods that keep Candida at bay – Eat organic food only, eat low to moderate sugar food in moderation (berries, pineapple, apple, papaya, orange, kiwi, cherries, grapes etc), eat plenty of colorful vegetables, 2 to 4 ounces per serving of organic grass-fed animal protein, gluten free whole grains (millets, Quinoa, Oats, Buckwheat, Amaranth), add healthy fats (Olive oil, Flax oil, Hemp oil, Coconut oil, Ghee, fish oil, avocados), eat nuts and seeds in moderation, eat beans and legumes no more than 2 times per week, drink plenty of water, and incorporate herbs and spices mentioned in the table below.
- Eat fermented foods like sauerkraut, kimchi, kanji, kefir made of coconut or goat’s milk. You can also take a good quality probiotic. Probiotics contain beneficial bacteria that keep candida in check.
- Sweeteners that are safe to consume on Anti-Candida diet – Stevia, Monk Fruit, Xylitol (excess of it can cause laxative effects) and Erythritol. Erythritol is the preferred one.
- Additional Supplements support – Digestive enzymes, Betaine HCL plus pepsin, digestive bitters, OX Bile, serrapeptase as a biofilm disruptor and a good quality plant-based Multivitamin.
- Cleanse your Lymphatic system and skin – through lymphatic massage, brisk walking or swimming, deep breathing exercises, jumping on a trampoline, dry brushing, hot and cold shower, or going for saunas that involves sweating through pores of skin
- Detox your liver and support gallbladder – do this by drinking herbal teas like milk thistle, Ginger Tulsi tea, lemon water, dandelion tea, burdock root tea.
- Cleanse your kidneys and bloodstream – sip hibiscus tea for kidneys and/or, red clover tea for cleansing blood.
- Keep your bowels moving – must have at least one bowel movement per day. If you are constipated, remedies for constipation are – Triphala, senna leaves, oxy powder, magnesium citrate or enema.
- Stimulate your Vagus Nerve – by singing your favorite song, chanting a mantra like OM, gargling with warm water early morning, yoga, meditation and deep belly breathing exercises, splashing cold water on face, or taking cold shower.
- Kitchen herbs effective against Candida (Ref – 41)
Herb
Main Biological Compound
Notable Effects on Candida
Clove
Eugenol
Antifungal, gut protection
Cinnamon
Cinnamaldehyde and Eugenol
Inhibits candida growth, anti-inflammatory
Turmeric
Curcumin
Disrupts membrane and mitochondria
Ginger
Phenolic acids, gingerols, paradols and shogaols
Antioxidant, antimicrobial, anti-diabetic, neuro- protective, analgesic, cardiovascular, gastrointestinal, anti-inflammatory, anticancer and antihypertensive.
Garlic
Ajoene, Allicin
Kill yeast cells
Oregano
Carvacrol, Thymol
Disrupts cell wall, biofilms
Thyme
Carvacrol, thymol and phenols
Rich in antioxidants and contains antiseptic and antifungal characteristics
Fennel
Phenolic compounds
Antifungal, Antioxidant, antimicrobial and anti-inflammatory
Chamomile
Flavonoids, terpenoids, phenolic compounds, apigenin and matricin
Antioxidant, anti-fungal, anti-inflammatory, anticancer, analgesic, anti-hypoglycemic, anti-stress and hepatoprotective.
Rosemary
1,8-cineole, α-pinene, carnosic acid, and rosmarinic acid
Rich in antioxidants that fight inflammation and fungus. Reduces biofilm formation and disrupting cell membranes
Role of Probiotics
Since Candida overgrowth mainly occurs due to local dysbiosis, antibiotic induced dysbiosis and immune deficiencies, it is reasonable to approach them with probiotics (28). This helps in reestablishing the local flora, which can compete against Candida colonization and infection.
Probiotics are “live microorganisms which, when consumed in adequate amounts, administer beneficial bacteria and confer health benefits on the host.”
Probiotics are often used in restoring gut flora in gastrointestinal diseases, but they can also be used for improving the health of other organs and systems, such as in case of candida – respiratory tract (28,32), the skin (28,33), the urogenital tract (28,34), and the oral cavity (28,38).
The most used probiotics belong to Lactobacillus spp., Bacillus spp., Bifidobacterium spp., and Saccharomyces spp. (28,31) that have shown effectiveness against Candida.
- Lactobacillus species – create an environment hostile to the growth of candida with fungitoxic effects and growth inhibition. Also, induce anti-adhesive/antibiofilm activities and antimicrobial activities proved on Candida Albicans and other species. In addition, cause alteration of the integrity of the fungal cell membrane and the fungal growth thus leading to fungal death (28).
- Bacillus species – like Bacillus subtilis – cause co-aggregation with candida species, anti-adhesive activity on planktonic Candida and inhibition and disruption of the fungal biofilm, antimicrobial activity proved on C.albicans, C.tropicalis, and C.krusei; induce protective action on mucosal integrity, prevention of the adhesion of C. albicans on medical devices and reduction of biofilm formation (28).
Furthermore, Probiotics preparations based on B. Clausii strains are widely diffused in the market to treat diarrhea and gut dysbiosis. Also, B. Clausii can produce riboflavin (vitamin B2), an essential vitamin whose deficiencies in humans are associated with pathologic status sustaining various diseases (28,35).
- Bifidobacterium species – Bifidobacterium Infantis, Bifidobacterium lactis – in a study, both probiotics guaranteed a statistically significantly higher overall survival and inhibited the incidence of systemic candidiasis. (28,36).
- Saccharomyces species – S. Boulardii and S. Cerevisiae – lead to stimulation of the host immune system against fungal infections – by downregulating the fungal induced inflammation and, increasing the production of protective IgA, IgG and IgM. The antifungal properties of S. cerevisiae have been proven both in mice with oropharyngeal candidiasis (28,37) and with vaginal candidiasis (28), where, after local administration, it was able to decrease the levels of C. albicans. S. Boulardii produces caprylic acid and other small bioactive molecules that are responsible for fungicidal effects.
Role of pharmaceutical antifungals
Candidiasis poses a severe clinical problem, but there are only three frontline antifungal drug classes for treatment: polyenes, echinocandins, and azoles, and each of them has disadvantages, such as severe side effects, limited antifungal spectrum, and antifungal resistance, that limit their clinical application (27,46).
Because these biofilms are resistant to conventional antifungals, the host immune system, and other environmental factors; It becomes significantly challenging to get rid of yeast infections.
The infection is conventionally eradicated with 3 types of antifungal drugs (28,46), mainly:
- Polyene Antifungal Drugs – include, Amphotericin B, Nystatin, and Pimaricin. These interact with sterols in the cell membrane (ergosterol in fungi, cholesterol in humans) to form channels through which small molecules leak from the inside of the fungal cell to the outside (39).
- Azole Antifungal Drugs – include, Fluconazole, Itraconazole, and Ketoconazole. These inhibit cytochrome P450-dependent enzymes (particularly C14-demethylase) involved in the biosynthesis of ergosterol, which is required for fungal cell membrane structure and function (39). Fluconazole is the main drug used in candiduric adults and in neonates (newborns). Oral fluconazole therapy gives bioavailability of 90%, diffuses readily into all body sites, and is concentrated in the urine and skin (39).
- Synthetic drugs derived from echinocandins – Caspofungin, micafungin, anidulafungin and rezafungin are semi-synthetic cyclic lipopeptides. The action of echinocandins is based on the inhibition of β-(1,3)-D-glucan synthesis that builds the fungal cell wall. These antibiotics also act on biofilm-forming yeasts especially on the Candida genus (40). Relative to Candida species, echinocandins exhibit fungicidal activity manifested by significant cell enlargement and distortion, which contributes to inhibition of cell proliferation. Echinocandins are recommended as a treatment for patients suffering from an invasive infection caused mainly by Candida, Aspergillus species and some other pathogenic fungi (28,40).
Limitations of antifungal drugs – While echinocandins have direct effects on the fungal wall, polyenes and azoles, in addition to altering the fungal plasma membrane by interfering with the synthesis and positioning of ergosterol, can also interfere with the cholesterol synthesis of the eukaryotic cell wall, thus presenting hepatotoxic (liver toxicity) and nephrotoxic (Kidney Toxicity) effects on the host. These side effects and drug resistance phenomena must not be underestimated in prolonged or recurrent use of these drugs (28,39,40,46).
References
- Nobile CJ, Johnson AD. Candida albicans Biofilms and Human Disease. Annu Rev Microbiol. 2015;69:71-92. doi: 10.1146/annurev-micro-091014-104330. PMID: 26488273; PMCID: PMC4930275.
- Hellstein JW, Marek CL. Candidiasis: Red and White Manifestations in the Oral Cavity. Head Neck Pathol. 2019 Mar;13(1):25-32. doi: 10.1007/s12105-019-01004-6. Epub 2019 Jan 29. PMID: 30693459; PMCID: PMC6405794.
- Medical Microbiology. 4th edition. Thomas J. Walsh and Dennis M. Dixon. Chapter 74, Disease Mechanisms of Fungi
- Medical Microbiology. 4th edition. Thomas J. Walsh and Dennis M. Dixon. Chapter 75, Spectrum of Mycoses
- The Candida Cure Book by Ann Boroch, CNC – The 90-day Program to balance your gut, beat candida, and restore vibrant health
- Gulati M, Nobile CJ. Candida albicans biofilms: development, regulation, and molecular mechanisms. Microbes Infect. 2016 May;18(5):310-21. doi: 10.1016/j.micinf.2016.01.002. Epub 2016 Jan 22. PMID: 26806384; PMCID: PMC4860025.
- Achkar JM, Fries BC. Candida infections of the genitourinary tract. Clin Microbiol Rev. 2010 Apr;23(2):253-73. doi: 10.1128/CMR.00076-09. PMID: 20375352; PMCID: PMC2863365.
- Jeanmonod R, Chippa V, Jeanmonod D. Vaginal Candidiasis. [Updated 2024 Feb 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK459317/
- Achkar JM, Fries BC. Candida infections of the genitourinary tract. Clin Microbiol Rev. 2010 Apr;23(2):253-73. [PMC free article] [PubMed]
- Martin Lopez JE. Candidiasis (vulvovaginal). BMJ Clin Evid. 2015 Mar 16;2015 [PMC free article] [PubMed]
- Makanjuola O, Bongomin F, Fayemiwo SA. An Update on the Roles of Non-albicans Candida Species in Vulvovaginitis. J Fungi (Basel). 2018 Oct 31;4(4) [PMC free article] [PubMed]
- Eckert, L. O. 2006. Acute vulvovaginitis. N. Engl. J. Med. 355:1244-1252. [DOI] [PubMed] [Google Scholar]
- Abdullah, A. N., S. M. Drake, A. A. Wade, and M. Walzman. 1992. Balanitis (balanoposthitis) in patients attending a department of genitourinary medicine. Int. J. STD AIDS. 3:128-129. [DOI] [PubMed] [Google Scholar]
- Dockerty, W. G., and C. Sonnex. 1995. Candidal balano-posthitis: a study of diagnostic methods. Genitourin. Med. 71:407-409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lisboa, C., A. Ferreira, C. Resende, and A. G. Rodrigues. 2009. Infectious balanoposthitis: management, clinical and laboratory features. Int. J. Dermatol. 48:121-124. [DOI] [PubMed] [Google Scholar]
- Robertson KD, Nagra N, Mehta D. Esophageal Candidiasis. [Updated 2023 Jul 31]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK537268/
- Rosołowski M, Kierzkiewicz M. Etiology, diagnosis and treatment of infectious esophagitis. Prz Gastroenterol. 2013;8(6):333-7. [PMC free article] [PubMed]
- Alsomali MI, Arnold MA, Frankel WL, Graham RP, Hart PA, Lam-Himlin DM, Naini BV, Voltaggio L, Arnold CA. Challenges to “Classic” Esophageal Candidiasis: Looks Are Usually Deceiving. Am J Clin Pathol. 2017 Jan 01;147(1):33-42. [PubMed]
- Takahashi Y, Nagata N, Shimbo T, Nishijima T, Watanabe K, Aoki T, Sekine K, Okubo H, Watanabe K, Sakurai T, Yokoi C, Kobayakawa M, Yazaki H, Teruya K, Gatanaga H, Kikuchi Y, Mine S, Igari T, Takahashi Y, Mimori A, Oka S, Akiyama J, Uemura N. Long-Term Trends in Esophageal Candidiasis Prevalence and Associated Risk Factors with or without HIV Infection: Lessons from an Endoscopic Study of 80,219 Patients. PLoS One. 2015;10(7):e0133589. [PMC free article] [PubMed]
- Choi JH, Lee CG, Lim YJ, Kang HW, Lim CY, Choi JS. Prevalence and risk factors of esophageal candidiasis in healthy individuals: a single center experience in Korea. Yonsei Med J. 2013 Jan 01;54(1):160-5. [PMC free article] [PubMed]
- Taylor M, Brizuela M, Raja A. Oral Candidiasis. [Updated 2023 Jul 4]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK545282/
- Millsop JW, Fazel N. Oral candidiasis. Clin Dermatol. 2016 Jul-Aug;34(4):487-94. [PubMed]
- Akpan A, Morgan R. Oral candidiasis. Postgrad Med J. 2002 Aug;78(922):455-9. [PMC free article] [PubMed]
- Hellstein JW, Marek CL. Candidiasis: Red and White Manifestations in the Oral Cavity. Head Neck Pathol. 2019 Mar;13(1):25-32. [PMC free article] [PubMed]
- Renga G, Bellet MM, Stincardini C, Pariano M, Oikonomou V, Villella VR, Brancorsini S, Clerici C, Romani L, Costantini C. To Be or Not to Be a Pathogen: Candida albicans and Celiac Disease. Front Immunol. 2019 Dec 5; 10:2844. doi: 10.3389/fimmu.2019.02844. PMID: 31867008; PMCID: PMC6906151.
- Kumamoto CA. Inflammation and gastrointestinal Candida colonization. Curr Opin Microbiol. 2011 Aug;14(4):386-91. doi: 10.1016/j.mib.2011.07.015. Epub 2011 Jul 28. PMID: 21802979; PMCID: PMC3163673.
- Lu H, Hong T, Jiang Y, Whiteway M, Zhang S. Candidiasis: From cutaneous to systemic, new perspectives of potential targets and therapeutic strategies. Adv Drug Deliv Rev. 2023 Aug; 199:114960. doi: 10.1016/j.addr.2023.114960. Epub 2023 Jun 10. PMID: 37307922.
- Maria Contaldo. Use of Probiotics for Oral Candidiasis: State of the Art and Perspective. A Further Step Toward Personalized Medicine?. Front. Biosci. (Elite Ed) 2023, 15(1), 6. https://doi.org/10.31083/j.fbe1501006
- Epstein JB, Pearsall NN, Truelove EL. Quantitative relationships between Candida albicans in saliva and the clinical status of human subjects. Journal of Clinical Microbiology. 1980; 12: 475–476. Cited within: 1 | Google Scholar | PubMed | Crossref
- Akpan A, Morgan R. Oral candidiasis. Postgraduate Medical Journal. 2002; 78: 455–459. Cited within: 1 | Google Scholar | PubMed | Crossref
- Mundula T, Ricci F, Barbetta B, Baccini M, Amedei A. Effect of Probiotics on Oral Candidiasis: A Systematic Review and Meta-Analysis. Nutrients. 2019; 11: 2449. Cited within: 3 | Google Scholar | PubMed | Crossref
- Campanella V, Syed J, Santacroce L, Saini R, Ballini A, Inchingolo F. Oral probiotics influence oral and respiratory tract infections in pediatric population: a randomized double-blinded placebo-controlled pilot study. European Review for Medical and Pharmacological Sciences. 2018; 22: 8034–8041. Cited within: 1| Google Scholar | PubMed | Crossref
- Sodré CS, Vieira MS, Estefan JL, Moraes C, Cavalcante FS, Dos Santos KRN, et al. The effect of probiotics on the clinical status of adult patients with atopic dermatitis: a systematic review. European Journal of Medical Research. 2022; 27: 94. Cited within: 1| Google Scholar | Crossref
- Ballini A, Santacroce L, Cantore S, Bottalico L, Dipalma G, Vito DD, et al. Probiotics Improve Urogenital Health in Women. Open Access Macedonian Journal of Medical Sciences. 2018; 6: 1845–1850. Cited within: 1| Google Scholar | Crossref
- Ghelardi E, Abreu Y Abreu AT, Marzet CB, Álvarez Calatayud G, Perez M Moschione Castro AP, et al. Current Progress and Future Perspectives on the Use of Bacillus clausii. Microorganisms. 2022; 10: 1246. Cited within: 1| Google Scholar | Crossref
- Wagner RD, Warner T, Pierson C, Roberts L, Farmer J, Dohnalek M, et al. Biotherapeutic effects of Bifidobacterium spp. on orogastric and systemic candidiasis in immunodeficient mice. Revista Iberoamericana De Micologia. 1998; 15: 265–270. Cited within: 3 | Google Scholar | Crossref
- Roselletti E, Sabbatini S, Ballet N, Perito S, Pericolini E, Blasi E, et al. Saccharomyces cerevisiae CNCM I-3856 as a New Therapeutic Agent Against Oropharyngeal Candidiasis. Frontiers in Microbiology. 2019; 10: 1469. Cited within: 3 | Google Scholar | Crossref
- Inchingolo F, Dipalma G, Cirulli N, Cantore S, Saini RS, Altini V, et al. Microbiological results of improvement in periodontal condition by administration of oral probiotics. Journal of Biological Regulators and Homeostatic Agents. 2018; 32: 1323–1328.Cited within: 1 | Google Scholar | Crossref
- Dixon DM, Walsh TJ. Antifungal Agents. In: Baron S, editor. Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston; 1996. Chapter 76. Available from: https://www.ncbi.nlm.nih.gov/books/NBK8263/
- Szymański M, Chmielewska S, Czyżewska U, Malinowska M, Tylicki A. Echinocandins – structure, mechanism of action and use in antifungal therapy. J Enzyme Inhib Med Chem. 2022 Dec;37(1):876-894. doi: 10.1080/14756366.2022.2050224. PMID: 35296203; PMCID: PMC8933026.
- Parham S, Kharazi AZ, Bakhsheshi-Rad HR, Nur H, Ismail AF, Sharif S, RamaKrishna S, Berto F. Antioxidant, Antimicrobial and Antiviral Properties of Herbal Materials. Antioxidants (Basel). 2020 Dec 21;9(12):1309. doi: 10.3390/antiox9121309. PMID: 33371338; PMCID: PMC7767362.
- Fox EP, Nobile CJ. The role of Candida albicans biofilms in human disease. In: Dietrich LA, Friedmann TS, editors. Candida albicans symptoms, causes and treatment options. Nova Science Publishers; 2013. pp. 1–24. [Google Scholar]
- Kojic EM, Darouiche RO. Candida infections of medical devices. Clin Microbiol Rev. 2004;17:255–267. doi: 10.1128/CMR.17.2.255-267.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox EP, Singh-babak SD, Hartooni N, Nobile CJ. Biofilms and antifungal resistance. In: Coste AT, Vandeputte P, editors. Antifungals from genomics to resistance and the development of novel agents. Caister Academic Press; 2015. pp. 71–90. [Google Scholar]
- Andes DR, Safdar N, Baddley JW, Playford G, Reboli AC, Rex JH, et al. Impact of treatment strategy on outcomes in patients with candidemia and other forms of invasive candidiasis: a patient-level quantitative review of randomized trials. Clin Infect Dis. 2012;54:1110–1122. doi: 10.1093/cid/cis021. [DOI] [PubMed] [Google Scholar]
- Spampinato C, Leonardi D. Candida infections, causes, targets, and resistance mechanisms: traditional and alternative antifungal agents. Biomed Res Int. 2013;2013:204237. doi: 10.1155/2013/204237. Epub 2013 Jun 26. PMID: 23878798; PMCID: PMC3708393.
- Rosenbach A, Dignard D, Pierce JV, Whiteway M, Kumamoto CA. Adaptations of Candida albicans for growth in the mammalian intestinal tract. Eukaryot Cell. 2010;9:1075–1086. doi: 10.1128/EC.00034-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vu B, Essmann M, Larsen B. Sodium choleate (NaCho) effects on Candida albicans: implications for its role as a gastrointestinal tract inhabitant. Mycopathologia. 2010;169:183–191. doi: 10.1007/s11046-009-9248-y. [DOI] [PubMed] [Google Scholar]
- Corouge M, Loridant S, Fradin C, Salleron J, Damiens S, Moragues MD, Souplet V, Jouault T, Robert R, Dubucquoi S, Sendid B, Colombel JF, Poulain D. Humoral immunity links Candida albicans infection and celiac disease. PLoS One. 2015 Mar 20;10(3):e0121776. doi: 10.1371/journal.pone.0121776. PMID: 25793717; PMCID: PMC4368562.
- Renga G, Bellet MM, Stincardini C, Pariano M, Oikonomou V, Villella VR, Brancorsini S, Clerici C, Romani L, Costantini C. To Be or Not to Be a Pathogen: Candida albicans and Celiac Disease. Front Immunol. 2019 Dec 5;10:2844. doi: 10.3389/fimmu.2019.02844. PMID: 31867008; PMCID: PMC6906151.
- Fourie R, Kuloyo OO, Mochochoko BM, Albertyn J, Pohl CH. Iron at the Centre of Candida albicans Interactions. Front Cell Infect Microbiol. 2018 Jun 5;8:185. doi: 10.3389/fcimb.2018.00185. PMID: 29922600; PMCID: PMC5996042.
- Arekar T, Katikaneni D, Kasem S, Desai D, Acharya T, Cole A, Khodayari N, Vaulont S, Hube B, Nemeth E, Drakesmith A, Lionakis MS, Mehrad B, Scindia Y. Essential role of hepcidin in host resistance to disseminated candidiasis. Cell Rep. 2025 May 27;44(5):115649. doi: 10.1016/j.celrep.2025.115649. Epub 2025 May 5. PMID: 40333187; PMCID: PMC12172023.
- McKie A. T., Barrow D., Latunde-Dada G. O., Rolfs A., Sager G., Mudaly M., et al. (2001). An iron-regulated ferric reductase associated with the absorption of dietary iron. Science 291, 1755–1759. 10.1126/science.1057206 [DOI] [PubMed] [Google Scholar][Ref list]
- Nemeth E., Tuttle M. S., Powelson J., Vaughn M. B., Donovan A., Ward D. M., et al. (2004). Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306, 2090–2093. 10.1126/science.1104742 [DOI] [PubMed] [Google Scholar][Ref list]
- Vylkova S, Carman AJ, Danhof HA, Collette JR, Zhou H, Lorenz MC. The fungal pathogen Candida albicans autoinduces hyphal morphogenesis by raising extracellular pH. mBio. 2011 May 17;2(3):e00055-11. doi: 10.1128/mBio.00055-11. PMID: 21586647; PMCID: PMC3101780.
- Ramos-Pardo A, Castro-Álvarez R, Quindós G, Eraso E, Sevillano E, Kaberdin VR. Assessing pH-dependent activities of virulence factors secreted by Candida albicans. Microbiologyopen. 2023 Feb;12(1):e1342. doi: 10.1002/mbo3.1342. PMID: 36825882; PMCID: PMC9808488.
- De Bernardis F, Mühlschlegel FA, Cassone A, Fonzi WA. The pH of the host niche controls gene expression in and virulence of Candida albicans. Infect Immun. 1998 Jul;66(7):3317-25. doi: 10.1128/IAI.66.7.3317-3325.1998. PMID: 9632601; PMCID: PMC108348.